 |
|||||||||||||||||
|
|||||||||||||||||
|
Self-Replicating Nucleic Acids
A prerequisite for constructing a protocell is supplying a mechanism for replicating the nucleic acids that fill structural, functional or informational roles. Two approaches under active investigation in our laboratory are the in vitro selection of ribozyme polymerases (replicases) and the non-enzymatic replication of nucleic acids using chemically-activated nucleotides. In both cases, the goal is a high-fidelity system for replicating essentially any nucleotide sequence.
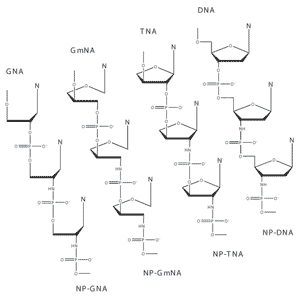
Non-enzymatic replication utilizes chemically-activated monomers or condensing agents to effect polymerization of monomers on template sequences. Chemical activation requires substituting a preferred leaving group in place of one of the hydroxyl groups of the 5´phosphate in a nucleoside monophosphate. Although this chemistry is well established in the literature, it has been limited by the slow reaction rates of naturally-occurring 3´OH-terminated nucleotides. We have recently found that substitution of the 3´OH with an amino group dramatically increases the reaction rate with activated monomers. The product, phosphoramidate DNA, is a well-known polymer with properties similar to naturally-occurring DNA and RNA. We are currently synthesizing a variety of chemically-activated nucleotides with 3´NH2 substitutions and are evaluating the kinetics and fidelity of non-enzymatic polymerization. |
|||||||||||||||||
|
|
|||||||||||||||||
| Copyright © 2006-2012 The Massachusetts General Hospital | |||


 Ribozyme polymerases are being selected from fully random or partially structured libraries of RNA or by modification of natural ribozymes with reversible phosphodiesterase bond cleaving activity.
Ribozyme polymerases are being selected from fully random or partially structured libraries of RNA or by modification of natural ribozymes with reversible phosphodiesterase bond cleaving activity.